Stable α-helices typically end with a charged amino acid to neutralize the dipole moment. In addition, the lack of a hydrogen on Proline's nitrogen prevents it from participating in hydrogen bonding.Īnother factor affecting α-helix stability is the total dipole moment of the entire helix due to individual dipoles of the C=O groups involved in hydrogen bonding. Proline also destabilizes α-helices because of its irregular geometry its R-group bonds back to the nitrogen of the amide group, which causes steric hindrance. Amino acids whose R-groups are too large (tryptophan, tyrosine) or too small (glycine) destabilize α-helices. The structural integrity of an α-helix is in part dependent on correct steric configuration. This regular pattern gives the α-helix very definite features with regards to the thickness of the coil and the length of each complete turn along the helix axis. Amino Acid 3-Letter Code 1-Letter Code Molecular Weight 1 Hydropathy 2 Alanine: Ala: A: 89.09: 6.3: Cysteine: Cys: C: 121.16: 7.0: Aspartate: Asp: D: 133.10: 1. Such a hydrogen bond is formed exactly every 4 amino acid residues, and every complete turn of the helix is only 3.6 amino acid residues. 
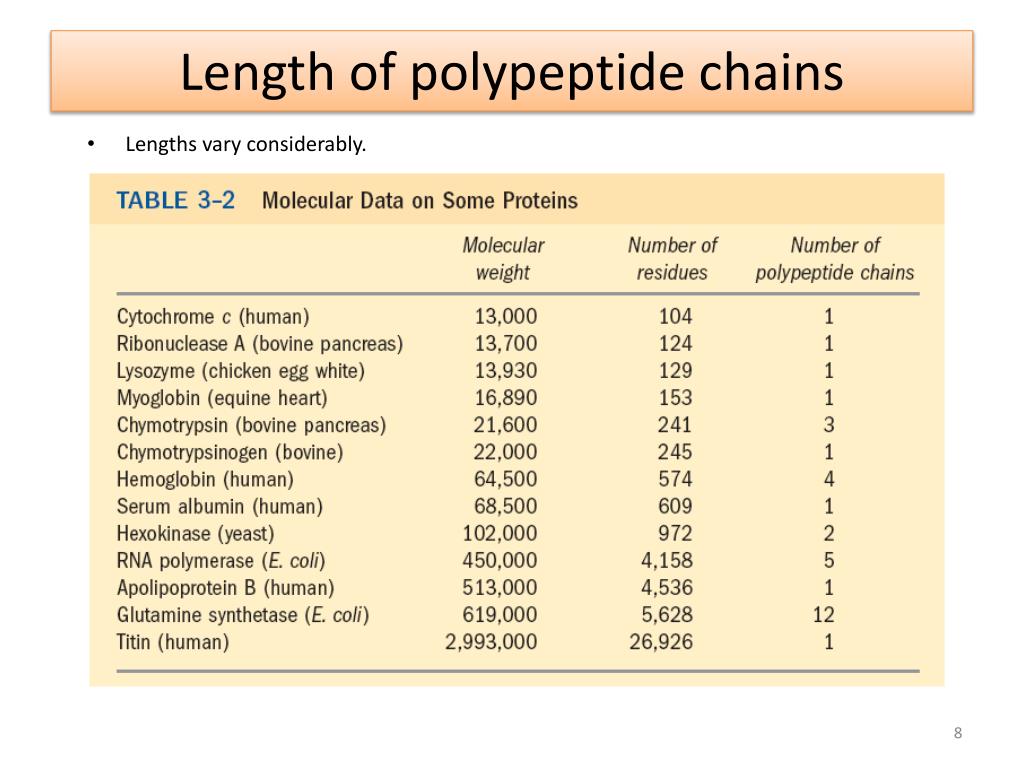
A protein contains one or more polypeptides. A peptide is two or more amino acids joined together by peptide bonds, and a polypeptide is a chain of many amino acids. This coil is held together by hydrogen bonds between the oxygen of C=O on top coil and the hydrogen of N-H on the bottom coil. A peptide bond forms when the amino group of one amino acid bonds to the carboxyl group of another amino acid. \( \newcommand\)Īn α-helix is a right-handed coil of amino-acid residues on a polypeptide chain, typically ranging between 4 and 40 residues.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
